A covalent bond is a form of chemical bonding that is characterized by the sharing of pairs of electrons between atoms, and other covalent bonds. The stable balance of attractive and repulsive forces between atoms when they share electrons is known as covalent bonding.[1]
Covalent bonding includes many kinds of interaction, including ?-bonding, ?-bonding, metal-to-metal bonding, agostic interactions, and three-center two-electron bonds.[2][3] The term covalent bond dates from 1939.[4] The prefix co- means jointly, associated in action, partnered to a lesser degree, etc.; thus a “co-valent bond”, in essence, means that the atoms share “valence”, such as is discussed in valence bond theory. In the molecule H2, the hydrogen atoms share the two electrons via covalent bonding. Covalency is greatest between atoms of similar electronegativities. Thus, covalent bonding does not necessarily require the two atoms be of the same elements, only that they be of comparable electronegativity. Although covalent bonding entails sharing of electrons, it is not necessarily delocalized. Furthermore, in contrast to electrostatic interactions (“ionic bonds”) the strength of covalent bond depends on the angular relation between atoms in polyatomic molecules.
en.wikipedia.org/wiki/Covalent_bond



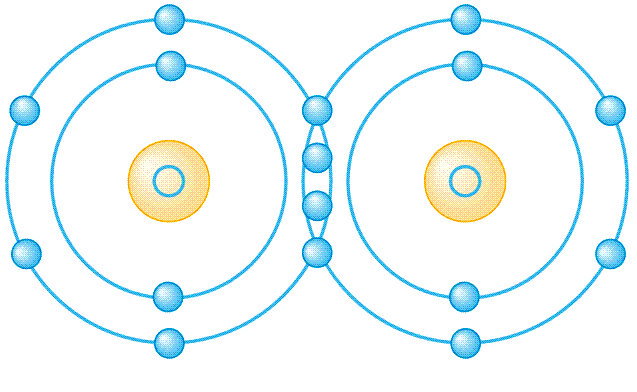

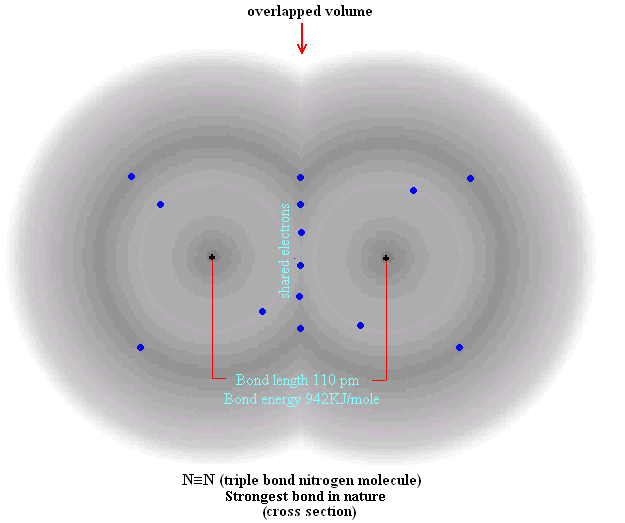
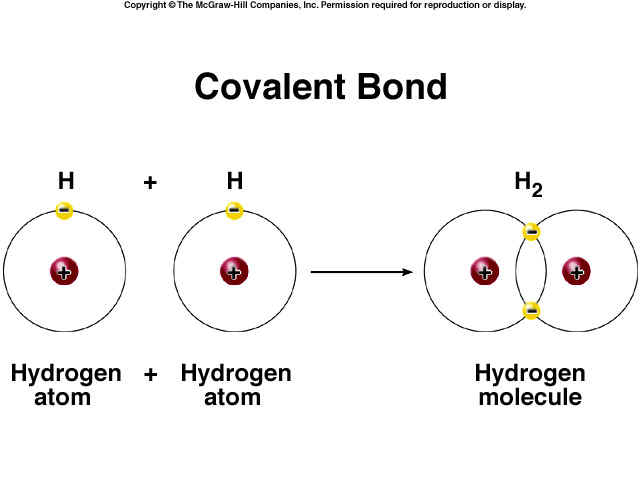





electron dot diagrams are ridiculously inaccurate. electrons exist in geometrically fixed orbitals. look up ‘f-orbitals’ for some really interesting ones.
I see boobs.